Hope for patients with cutaneous squamous cell carcinoma (CSCC): Cemiplimab-Rwlc (Libtayo)
Last Updated on October 30, 2018 by Joseph Gut – thasso
October 31, 2018 – The American Food and Drug Administration (FDA) recently approved Cemiplimab-Rwlc (Libtayo) injection for intravenous use for the treatment of patients with metastatic cutaneous squamous cell carcinoma (CSCC) or locally advanced CSCC who are not candidates for curative surgery or curative radiation. Cemiplimab-Rwlc (Libtayo) is the first FDA approval of a drug specifically targeting advanced CSCC.

Cemiplimab-Rwlc (Libtayo) works by targeting the cellular pathway known as PD-1 (protein found on the body’s immune cells and some cancer cells). By blocking this pathway, the drug may help the body’s immune system fight the cancer cells. These type of drugs are generally called immune checkpoint inhibitors (ICIs).
Cemiplimab-Rwlc (Libtayo) continues a shift in oncology toward identifying and developing drugs aimed at specific molecular tumor target. With the approval of Cemiplimab-Rwlc (Libtayo), the FDA has approved until now six ICIs targeting the the PD-1 / PD-L1 pathway for treating a variety of tumors, from bladder to head and neck cancer, and now advanced CSCC.
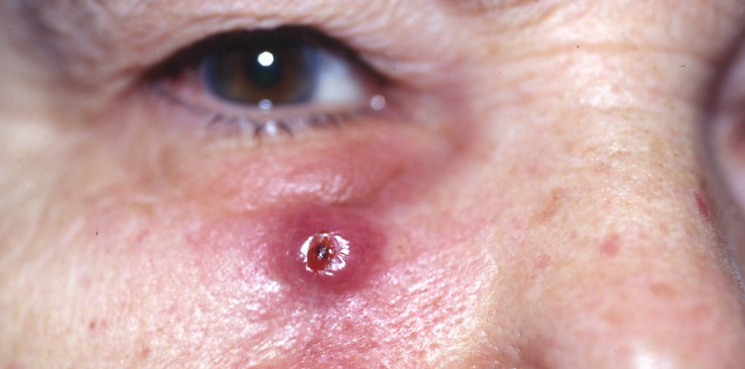
CSCC is the second most common human cancer in the United States with an estimated annual incidence of approximately 700,000 cases. The most common form of skin cancer is basal cell cancer. Squamous cells are thin, flat cells that look like fish scales and are found in the tissue that forms the surface of the skin. CSCC usually develops in skin areas that have been regularly exposed to the sun or other forms of ultraviolet radiation. While the majority of patients with CSCC are cured with surgical resection, a small percentage of patients will develop advanced disease that no longer responds to local treatments including surgery and radiation. Advanced CSCC may cause disfigurement at the site of the tumor and local complications such as bleeding or infection, or it may spread (metastasize) to local lymph nodes, distant tissues and organs and become life-threatening.
The efficacy of Cemiplimab-Rwlc (Libtayo) was studied in two open label clinical trials. A total of 108 patients (75 with metastatic disease and 33 with locally-advanced disease) were included in the efficacy evaluation. The study’s primary endpoint was objective response rate, or the percentage of patients who experienced partial shrinkage or complete disappearance of their tumor(s) after treatment. Results showed that 47.2 percent of all patients treated with Cemiplimab-Rwlc (Libtayo) had their tumors shrink or disappear. The majority of these patients had ongoing responses at the time of data analysis.
Similar to all ICIs so far, Cemiplimab-Rwlc (Libtayo) does not come without a price, however. The safety of Cemiplimab-Rwlc (Libtayo) was examined in this clinical trials with somewhat limited number of patients. Common side effects of Cemiplimab-Rwlc (Libtayo) included fatigue, rash and diarrhea. However, Cemiplimab-Rwlc (Libtayo) may cause the immune system to attack normal organs and tissues in any area of the body and can affect the way they work. These reactions can sometimes become severe or life-threatening and can lead to death. These reactions include the risk of immune-mediated adverse reactions including lung problems (pneumonitis), intestinal problems (colitis), liver problems (hepatitis), hormone gland problems (endocrinopathies), skin (dermatologic) problems and kidney problems. Patients should also be monitored for infusion-related reactions. Cemiplimab-Rwlc (Libtayo) can cause harm to a developing fetus; women should be advised of the potential risk to the fetus and to use effective contraception.
Thasso Post had just recently an article about the potential deadly dangers for some rare patients when taking (or being prescribed) ICIs. This may be true for patients with an autoimmune predisposition, which may be reactivated through the mechanism of unlocking the PD-1 /PD-L1 pathway which is fundamentally involved in the regulation of autoimmunity.
See also this short introductory video on CSCC:

