FDA extends armamentarium to treat multiple myeloma: Ixazomib (Ninlaro) and Daratumumab (Darzalex) approved

Last Updated on November 22, 2015 by Joseph Gut – thasso
November 21, 2015 – During the last few days, the American Food and Drug Administration (FDA) just extended the armamentarium in the fight against multiple myeloma by two new entries, namely Ixazomib (Ninlaro) and Daratumumab (Darzalex). Both drugs are intended to treat those patients who have received at least one (Ixazomib (Ninlaro) or three (Daratumumab (Darzalex) prior therapies for multiple myeloma, and whose disease has become resistant to those prior treatment options.
Multiple myeloma is a form of blood cancer that occurs in infection-fighting plasma cells (a type of white blood cell) found in the bone marrow. These cancerous cells multiply, produce an abnormal protein and push out other healthy blood cells from the bone marrow. The disease may result in a weakened immune system
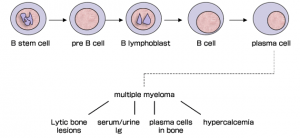
and cause other bone or kidney problems. The National Cancer Institute (NCI) estimates there will be this year alone 26,850 new cases of multiple myeloma and 11,240 related deaths in the US.
Of the two newly approved drugs, Ixazomib (Ninlaro) is a type of cancer drug called a proteasome inhibitor, having the 20S-proteasome subunit beta-5 (PSMB5) as molecular target, and working by blocking enzymes from multiple myeloma cells, hindering their ability to grow and survive. Ixazomib (Ninlaro) is the first oral proteasome inhibitor and is approved in combination with another FDA-approved treatments for multiple myeloma called Lenalidomide (Revlimid) and Dexamethasone (a type of corticosteroid). The other drug, Daratumumab (Darzalex), given as an infusion, is a monoclonal antibody that binds to CD38 and helps certain cells in the immune system attack cancer cells.
The efficacy of Ixazomib (Ninlaro) was demonstrated in an international, randomized, double-blind clinical trial of 722 patients whose multiple myeloma came back after, or did not respond to, previous treatment. Study participants received either Ixazomib (Ninlaro) in combination with lenalidomide and dexamethasone or placebo plus lenalidomide and dexamethasone. Those taking Ixazomib (Ninlaro) lived longer without their disease worsening (average 20.6 months) compared to participants taking the other regimen (14.7 months). In contrast, the efficacy of Daratumumab (Darzalex) was demonstrated in two open-label studies. In one study of 106 participants receiving Daratumumab (Darzalex), 29 percent of patients experienced a complete or partial reduction in their tumor burden, which lasted for an average of 7.4 months. In the second study of 42 participants receiving Daratumumab (Darzalex), 36 percent had a complete or partial reduction in their tumor burden.
Both come with adverse effects. The most common adverse effects of Ixazomib (Ninlaro) are diarrhea, constipation, low blood platelet count (thrombocytopenia), peripheral neuropathy (numbness and pain from nerve damage, usually in the hands and feet), nausea, peripheral edema (fluid under the skin causing swelling), vomiting and back pain, while the most common side effects of Darzalex were infusion-related reactions, fatigue, nausea, back pain, fever and cough. Darzalex may also result in low counts of infection-fighting white blood cells (lymphopenia, neutropenia, and leukopenia) or red blood cells (anemia) and low levels of blood platelets (thrombocytopenia). In addition, blood banks should be informed that patients are receiving Darzalex because the drug may interfere with certain tests that are done by blood banks (such as antibody screening) for patients who need a blood transfusion. Women who are pregnant should not use Darzalex, and women planning to become pregnant should use effective contraceptives during and for at least three months after treatment.


As of May 20, 2016, Daratumumab (Darzalex) is a cancer medicine used on its own to treat adults with multiple myeloma received the market authorisation valid throughout the European Union. See here for the EPAR: http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/human/medicines/004077/human_med_001979.jsp&mid=WC0b01ac058001d124